Healthcare
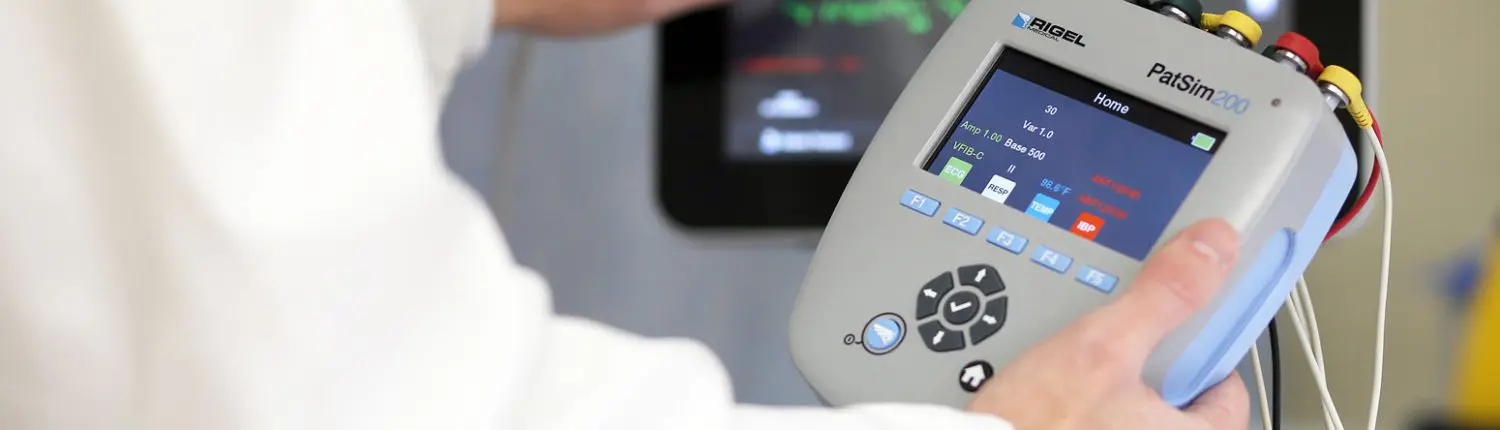
Healthcare test equipment refers to devices designed to test the safety, accuracy, and performance of medical equipment. Healthcare calibration plays a critical role in ensuring compliance with industry standards and patient safety. Commonly used by medical device manufacturers, private hospitals, healthcare providers, and medical device service companies, it is also essential for public sector health organisations, such as NHS EBME departments, and third-party maintenance providers within the medical device industry.